
Boone, J.L., C. Elaine Furbish, and Kent Turner. 1987. Control of Phragmites communis. Results of burning, cutting, and covering with plastic in a North Carolina salt marsh. School of forest Resources and Institute of Ecology, University of Georgia, CPSU Technical Report No. 41.
Note: this online version has been edited for grammar and syntax since the original publication. Figures and tables are at the end.
Control of Phragmites communis:
Results of Burning, Cutting, and Covering with Plastic in a North Carolina Salt Marsh
CPSU Technical Report 41
Jim Boone, Elaine Furbish, and Kent Turner
National Park Service
Cooperative Park Studies Unit
Institute of Ecology
University of Georgia
Athens, GA 30602
December 1987
James L. Boone
Elaine Furbish
School of Forest Resources and Institute of Ecology, Cooperative Park Studies Unit, University of Georgia, Athens, GA 30602
Kent Turner
Cape Hatteras National Seashore, Manteo, NC 27954
Abstract
Methods of controlling of Phragmites communis by manual cutting, burning, and covering with sheets of clear plastic were evaluated. None of these methods, used on a single occasion, was effective in killing the plant. There was a significant reduction in stem density in three of four plots that were either cut or burned, and then covered with plastic (p < 0.01; mean reduction = 49%). There were significant statistical reductions in stem heights, but these reductions may not be biologically significant because the stems were still growing when sampled. Cut and burned stems were an average of 21 percent shorter than they had been before treatment, and stems covered with plastic were 65 percent shorter.
There were mechanical problems associated with plastic. These problems included moving large sheets of plastic into the marsh, holding it in place in high winds, and its rapid degradation in sunlight. Covering with plastic may be a useful for killing Phragmites without herbicides if treatment application methods are modified. Recommendations are made, but they need to be evaluated.
Introduction
Phragmites communis Trin. (Radford et. al. 1968), or Common Reed, is a tall (to 4 m) perennial grass that grows in dense monotypic thickets (to 400 stems/m2) in wetlands. Phragmites spreads rhizomatously, can start from a single fragment of rhizome, and can spread to cover hundreds of hectares. Phragmites is of concern to wetland managers because it grows rapidly, excludes other plant species, provides little wildlife habitat, and is of little aesthetic or recreational value.
Because of these undesirable characteristics, many attempts have been made to control the spread of this plant, but with little success. Attempts to control Phragmites have included burning, cutting, draining ponds, flooding, disking, mowing, insect pests, and herbicides (Jones and Lehman 1987). These techniques show immediate effects, but herbicides are the only technique that is effective in the long term (Jones and Lehman 1987). Phragmites, however, typically grows in wetlands where the use of herbicides is difficult, if not impossible, due to regulations and environmental concerns.
The inability to use herbicides and the ineffectiveness of standard control methods have lead managers to look for alternatives. Heating the soil to temperatures lethal for plants by covering with sheets of clear plastic has been used to remove vegetation in marshes (unpublished MS). A pilot study using clear plastic to control Phragmites was conducted in 1986 and showed a reduction in stem density of about 85% (Furbish et. al. 1987). In this study, we quantify the cost and effectiveness of cutting, burning, and covering with plastic as control methods for P. communis.
Methods
The study design included two stem removal methods, cutting and burning, and two post-removal follow-up methods, plastic and herbicides. Each combination (cut, burn, cut-plastic, burn-plastic, cut-herbicide, and burn-herbicide) was to be applied to two treatment areas. However, not all of the burn treatments could be applied and no herbicides were used. In addition, a single stand was burned and cut repeatedly when the stems grew to 3-5 dm to see if the plants energy reserves could be drained. Repeat cutting was only conducted as a pilot study, and is included here because it showed promise.
Three stands of Phragmites were selected on Bodie Island, Cape Hatteras National Seashore, and two stands were selected on Pea Island National Wildlife Refuge (NWR) (Fig. 1, 2a, 2b). Site selection was based on size and location of the stand of Phragmites. Stand size was selected to be greater than 30 m by 15 m so that two non-adjacent 12x12-m plots could be placed in each stand, but not so big as to prevent treating the entire stand. In one case, four plots were placed in one stand. Each treatment covered approximately 500- to 600-m2. Sites were selected to be away from pond edges and near roads. Trenches were dug to cut rhizomes and separate the pairs of 12x12-m plots in each stand.
Cutting was conducted in April 1987 when the new stems were emerging. Manual removal of Phragmites was difficult and time consuming, but a short, heavy scythe blades on a metal handle was the most effective tool. Swing blades, hand clippers, axes, weed eaters, and other tools were tried, but none were effective. Cut stems were removed from the plots.
Burning was conducted on April 30th, 1987 when the marsh was still flooded. Phragmites at Cape Hatteras tends to grow on high ground (measured in centimeters of change from the surrounding area, by personal observation), so the base of the Phragmites plants can be dry while the surrounding grassy areas are wet. There must be dry ground litter present to ignite the Phragmites, but once started, fire will carry over wet areas. Burning was very easy and time efficient because fire lines were formed by walking through the marsh grasses (mostly Spartina patens) around the Phragmites, thereby wetting the grasses and creating a "wetline". The stands on Pea Island could not be burned because they were flooded.
Sheets of plastic were spread over the plots after cutting or burning. The plastic measured six-mil by 27.4 m by 12.2 m (90 by 40 feet) and weighed 51.8 kg (114 pounds). Each treatment required two sheets of plastic. The plastic was too heavy to carry into the marsh, and was airlifted by the U.S. Coast Guard. The plastic was difficult to anchor in place due to strong winds. The sheets were initially taped together with construction-grade duct tape, but the tape deteriorated after 2-4 days of exposure to sunlight and high temperatures. Wind then carried the plastic off the plots and pulled up the edges that were buried under 2-3 dm of soil. To hold the plastic in place, sheet edges were rolled together and weighed down with sandbags weighing 5 kg spaced 1 dm apart. In addition, sandbags were placed in a regular grid on the plastic about 2 m apart. Sandbags were made from wastebasket-sized plastic bags. With the edges secure, rainwater was helpful in holding the plastic in place. These measures created many small holes in the plastic, but the holes did not seem to interfere with heating.
Sampling design was 8 randomly selected 0.25-m2 quadrats in the 12-m2 plots and 5 fixed 1-m2 quadrats on the edge of the plot. Variables recorded per plot were stem height, stem density, maximum height, other species present, and amount of time spent working in the plot. Temperatures under the plastic were recorded independently of this sampling scheme.
Pre-treatment sampling was conducted in early April as new shoots were beginning to emerge. Post-treatment sampling was conducted in early September before the end of the growing season.
Results
The cutting treatment reduced all standing stems to less than 20- to 30-cm high. The burn treatment reduced all but a few stems to less than 10- to 20-cm in the plots where fire would carry. As long as the plastic was in place, it prevented sprouting. A nearly complete die-off of all species under the plastic was noted in only two to three days.
Under-plastic temperatures were recorded as high as 76°C (169°F) when ambient temperatures were 38°C (100°F). Under-plastic air temperatures averaged 59°C. Under-plastic temperatures averaged 33°C 5 cm beneath soil surface, and 28°C at 20 cm. This compares with mean temperature of 25°C and 23°C under standing Phragmites, and an average of 26°C and 24°C under various types of marsh vegetation.
Burning, cutting, and plastic caused significant reductions in stem height (p < .05). Burning and cutting had the same effect. Plastic had an additional effect that was approximately twice that of burning or cutting (Fig. 3 and 4). Cutting and burning had no significant effect on stem density, and the effects of plastic were mixed. On three plastic-covered plots, there was a significant reduction in stem density (p < .05); but on the fourth, there was an insignificant increase (Fig. 5 and 6).
All but one of the fixed plots on the edges of Stands 1, 2, and 3 showed an increase in stem density (p < 0.01). Because the fixed plots for Stands 4 and 5 (the control plots) were not relocatable, it is not known if this is a treatment result.
Multiple cutting after burning resulted in a reduction in the growth rate, but did not kill the plants (Fig. 7). In 55 days from late July to early September, the sprouts grew to only 20 to 30-cm, whereas sprouts released from plastic at the end of August grew to over 50 cm by early September. In addition, there was a nearly 100% ground cover of species other than Phragmites (mostly S. patens, also numerous herbs) growing amongst the Phragmites stems by the fourth cutting.
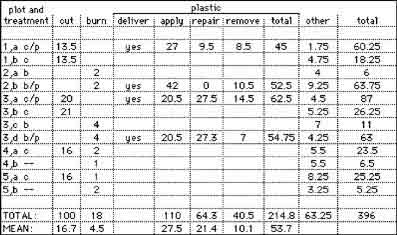
Cutting took an average of 17 hours per plot, burning took three hours per plot, and all phases of plastic took 53 hours per plot. The times spent in each treatment are summarized in Table 1. The hours from early in the project are correct totals per stand, but their distribution among plots was averaged.
Discussion
All treatments applied showed a negative effect on stem height and stem density. However, because these treatments were applied only once and a stand can regenerate from a single rhizome, these treatments were ineffective for killing the plants. In spite of this pessimistic analysis, the application of plastic and multiple cutting show promise.
There were statistically significant reductions in stem height and density, but there seemed to be a difference in the way these two parameters responded. It is likely that the reduction in stem height across all plots was simply the result of not letting them grow back before post-treatment sampling, and height may not be an important variable in evaluating treatment effectiveness. The slight decrease in stem density across the cut and burn plots may be the result of removing old, dead stems. The significant reduction in density on three of four plastic covered plots, however, may indicate a biological treatment effect.
Plants on the cut and burn plots had plenty of stored energy in the rhizomes to regenerate to essentially pre-treatment condition after treatment (Figures 3-7), and the plants continuously sent up new shoots under the plastic (personal observation). The reduction in stem density under the plastic may show a reduction in the ability of the plants to send up new shoots, and may show rhizome energy reserves being depleted. This is in accord with data from the repeated cutting where growth rates and densities decreased (personal observation).
The increase in stem density under plastic in Plot 2b may be related to the length of time that the plastic was on the plot. Time under plastic was shortest for this plot, about 40 days, and may correspond to the early dates on the multiple-cutting graph (Fig. 7), which showed an increase in plant growth rates early on. By comparison, the plastic was on Plots 1b, 3a, and 3d for about 70 days, corresponding to the peak of the graph on Figure 7. Because the plants were probably able to recover some of their energetic costs between cuttings in the multiple-cut plot, the graph of "regeneration effort" under plastic would probably shift to the left of that shown in Figure 7 because of a complete lack of return on the plant's "efforts" to regenerate. In the 1986 pilot study, plastic was on the plot for about 120 days and there was minimal resprouting, even in 1987, and the sprouts in that plot appeared to have grown in from the sides of the plots (personal observation). This suggests that plastic may be effective, but that there are minimum lengths of time required for it to drain the plant's energy reserves, thereby killing it.
The main problems with plastic involved moving it into the marsh, holding it in place, and deterioration due to sunlight. We found no advantage to using large sheets of plastic. Sheets 12x12 m (40x40 ft) would have been more manageable and would only weigh about 23 kg (50 pounds). We also found that there was no advantage to burying the edges of the plastic, as there was a complete kill of surface vegetation within 1 m of the edge of the unburied plastic. There is probably no reason to roll the edges of the plastic together where the sheets meet if they are overlapped. The plastic was held in place, even in strong winds, using a grid of sandbags spaced about 2-m apart.
The life spans of 4-mil (used in the pilot study) and 6-mil plastic are similar, roughly 2 months. This suggests that less expensive 4-mil plastic would probably work just as well. After 8-10 weeks in the sun, the plastic deteriorated. It is imperative to remove the plastic from the field before this time because it suddenly becomes brittle and fragments into thousands of tiny pieces. In Plot 3a, this resulted in many tiny pieces of plastic being left behind and greatly increased the time required to pick up those that could be removed. The short life of the plastic could be overcome. We took no measurements of under-plastic death rate, but there was a complete surface kill in only three or four days. If the plastic were rolled out for four or five days, then rolled up and stored away from direct sunlight (e.g., under black plastic) for two weeks, and rolled out again, it could be used to kill sprouts as they emerge. By storing the plastic out of the sun, it could probably be used for an entire season. This would then have the same effect as repeated cutting, but larger areas could be treated more effectively and in less time.
Much of the variation in time spent on plastic application (especially on Plot 2b) reflected the need to cut stems that had regrown. This time could be eliminated by applying plastic soon after burning or cutting.
If plastic is infeasible, then repeated cutting may be effective, but this cannot be answered until its effects can be determined during the 1988-growing season [ed. note: this was not done]. Cutting new stems (< 1-m high) was quick and relatively easy.
In summary, burning is the easiest and least expensive manual method for removing the old, dead stems before any post-removal follow-up treatment (if the cost of fireline construction can be minimized). Covering with plastic probably is an effective, non-herbicidal treatment, but the plastic must be left in place for longer than 70 days, but probably less that 120 days. Burning, followed by covering with plastic, was the most cost-effective method of manual control of Phragmites. If the plastic application methods were modified, this treatment combination would likely be more effective and less costly.
Acknowledgements
This project was supported by a National Park Service cooperative agreement with Rutgers University, and subcontracted to the University of Georgia. This study was a subproject of #10-21-RR271-187 RF Rutgers IST Bratton. Susan Bratton was principal investigator. The primary goal of the project was to investigate the possible use of fire to manage shrub thickets. This subproject assessed the impact of fire on Phragmites and potential means of controlling Phragmites spread if stimulated by fire. The subproject designed by Elaine Furbish, and Furbish coordinated all but the last two months of the study. Thanks are extended to Elizabeth LaRue for her help in the field and the office, to the U.S. Coast Guard for air support, to Sam Cooper, RoseMary Bauman-Barron, and Allen Barron for their help in the field.
Literature Cited
Furbish, C.E., S.P. Bratton, and K. Turner. 1987. Removal of Phragmites communis using clear plastic mulch. Restoration and Management Notes. 51(2). 1987.
Gallagher, J.L., et. al. 1987. Seasonal Patterns in Storage Carbohydrates in Phragmites australis. in Whitman, W.R. and W.H. Meredith (eds) 1987. Waterfowl and Wetlands Symposium: Proceeding of a Symposium on Waterfowl and Wetlands Management in the Coastal Zone of the Atlantic Flyway. Delaware Coastal Management Program, Delaware Department of Natural Resources and Environmental Control, Dover, Delaware. 522 pp.
Jones, W.L., and W.C. Lehman. 1987. Phragmites Control and Regeneration Following Aerial Application of Glyphosate in Delaware. ibid.
Radford, A.C., H.A. Ahles, and C.R. Bell. 1968. Manual of the Vascular Flora of the Carolinas. The University of North Carolina Press, Chapel Hill. 1183 pp.

Figure 1. Study areas on Bodie Island, Cape Hatteras National Seashore, and on Hatteras Island in Pea Island NWR.

Figure 2a. Study sites 1, 2, and 3 on Bodie Island.

Figure 2b. Study sites 4 and 5 on Pea Island NWR.
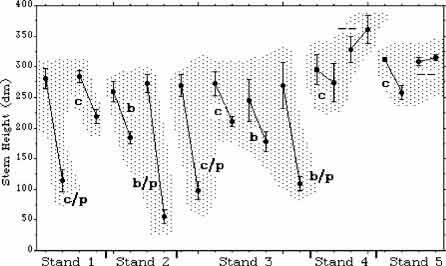
Figure 3. Pre- and post-treatment mean maximum heights per plot with 95% Error bars. In each case, the left member of each pair is pre-treatment (c = cut, b = burn, c/p = cut and plastic, b/p = burn and plastic, -- = no treatment).
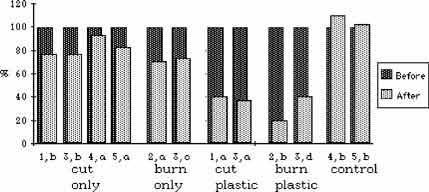
Figure 4. Differences in mean maximum stem heights before and after treatment. In each case, height is expressed as a percent where the pre-treatment height per plot is defined as 100%.
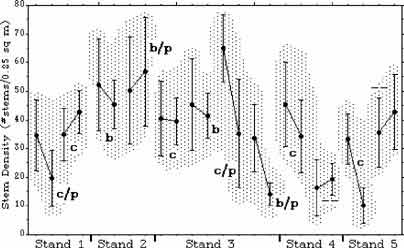
Figure 5. Pre- and post-treatment mean stem densities with 95% error bars. See Fig. 3 for explanation of symbols. Plot 5a (cut) represents a sampling anomaly.
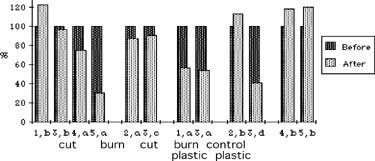
Figure 6. Differences in stem density. See Figure 4 for explanation of symbols. Plot 5a represents a data anomaly.

Figure 7. Change in growth rate after repeated cutting. Open circles represent the days on which the Phragmites was cut and measured.
Table 1. Hours spent working in the plots.
| Concerning Copyright | Home |